Advancing Medical Breakthroughs with Nanotechnology Solutions
POP Biotechnologies develops platform technologies addressing critical needs in oncology and infectious disease.
Our Expertise
The Power of PoP

POP BIO uses two patented porphyrin-phospholipid (PoP) liposome technologies to create nanomedicines to address needs in address needs in infectious disease, oncology and chronic diseases
Accelerated Vaccine Development
The Spontaneous Nanoliposome Antigen Particleization (POP BIO SNAP) platform enables the rapid development of highly immunogenic vaccines
Targeted Chemotherapy
Chemophototherapy (CPT) using PoP liposomes to deliver targeted concentrations of chemotherapy drugs to tumors expose to specific laser light.

Latest News

POP Biotechnologies to Receive up to US $9.7 Million in CEPI Funding to Advance SNAP™ Nanoparticle Vaccine Platform Into Phase 1 Clinical Testing in the United States
June 3, 2026
CEPI funding will support continued development of POP BIO’s rapid-response SNAP™ protein vaccine platform, including an H5N1 avian influenza vaccine candidate and future Disease X preparedness
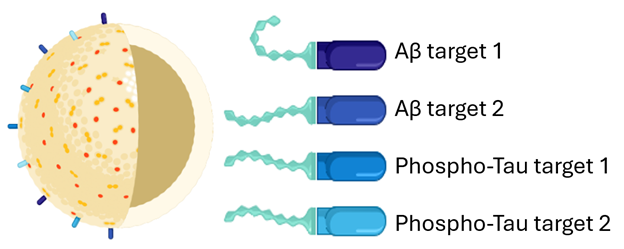
POP Biotechnologies Awarded $2.46M NIH Grant for Alzheimer's Immunotherapy
October 1, 2025
POP Biotechnologies, Inc. (POP BIO), has been awarded a $2.46M USD grant by the National Institutes of Health (NIH) to pursue development of a “mosaic” active immunotherapy against Alzheimer’s disease (AD).

POP BIO and CEPI announce collaboration for faster responses to Disease X
July 24, 2025
POP BIO announces award of $1.5M award from CEPI to advance SNAP platform into development of a vaccine against severe fever with thrombocytopenia syndrome (SFTS)
Discover Our Exciting Clinical Developments
Stay updated with our latest projects and collaborations in the field of biotechnology.
We are Actively Expanding Our Strong IP Protection
Exclusive license from SUNY for entire POP BIO SNAP and related patent portfolio
Formulations for MHC-I Restricted Epitope Immunization
Priority 1/2021
PCT WO2022155156
Particle based formulations of SAR-CoV-2 receptor binding domain
Priority 9/2020
PCT WO2022072654A1
Continuation In Part of above with examples in RSV, Influenza, and Lyme antigens
Priority 8/2019
U.S.A US20190255188
PCT WO2022072654A1
Serum-stable Compositions and Methods for Light Triggered Release of Materials
Priority 9/2015
European Union
Designated:
Belgium, France, Germany, United Kingdom,Italy, Ireland, Netherlands, Spain, Sweden, Switzerland EP3277264
Belgium, France, Germany, United Kingdom,Italy, Ireland, Netherlands, Spain, Sweden, Switzerland EP3277264
Nanostructures comprising cobalt porphyrin-phospholipid conjugates and polyhistidine-tags
Priority 4/2015
Australia AU2016248454
Canada CA2981359
China CN107708671
India 201717037629
Japan JP2018513156
U.S.A. US10,272,160
European Union
Belgium 167809961
France WO2020223395
Germany CA000002981359A1
United Kingdom EP16780996
Italy 502021000020480
Ireland 3283056
Netherlands EP3283056
Spain ES2865392
Sweden EP16780996.1
Switzerland 16780996
Compositions and Method for Light Triggered Release of Materials from Nanovesicles
Priority 12/2012
European Union
